Category: Parkinson's Disease: Non-Motor Symptoms
Objective: Visuomotor functions required for depth and dimensionality perception are significantly affected in a large number of patients with Parkinson’s disease (PD). Vergence is a type of eye movement contributing to this and is compromised in PD. Monocular blur and binocular disparity are important triggers for vergence and are controlled via different pathways. We asked which of the two vergence types is affected in PD.
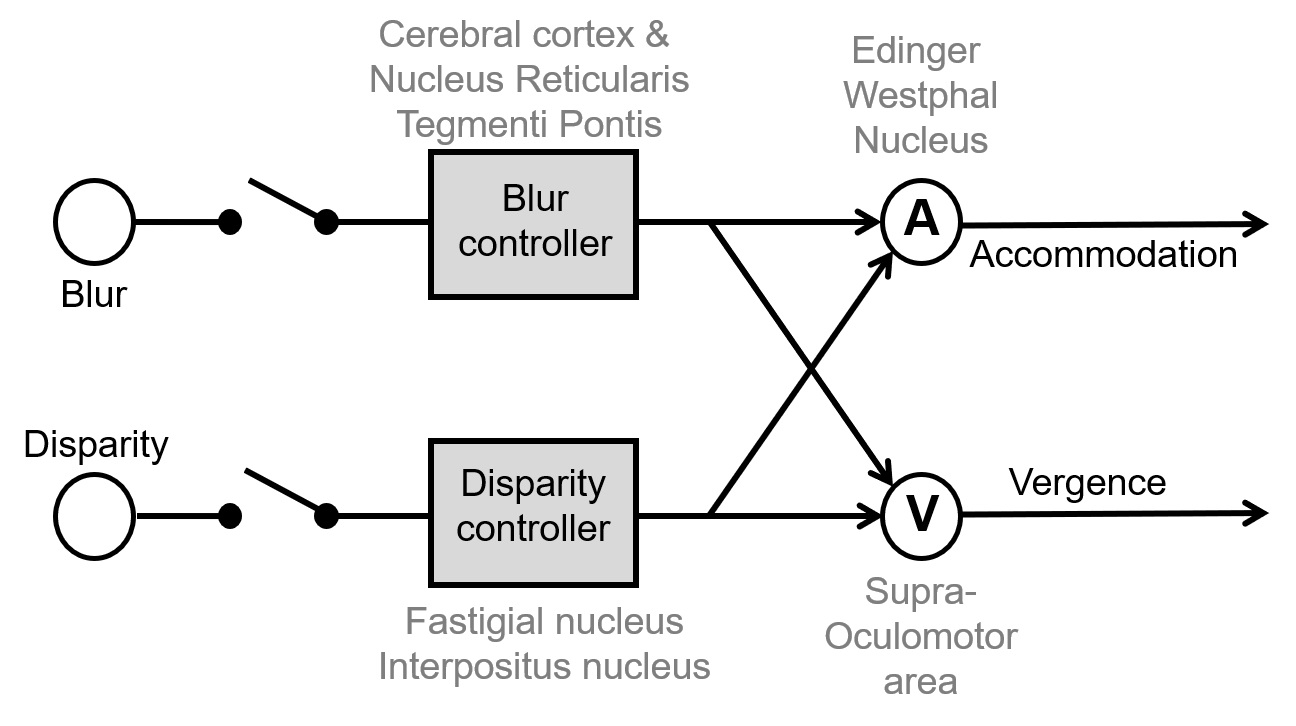
Background: Both convergence (far to near) and divergence (near to far) eye movements are associated with accommodation and are active players in depth perception. The two key sensory drives for vergence and accommodation, are disparity and blur.[1-4] Conceptually vergence and accommodation systems are cross-coupled, i.e. change in blur or disparity can trigger both accommodation as well as vergence, as they do when present together.[5] Fig 1 depicts simplified concept of cross-coupling between accommodation and vergence driven by blur (BD) and/or disparity (DD). We asked which aspect of the pathway depicted in Fig 1 is affected in PD.
[figure 1]
Method: Both vergence types were examined in 14 PD patients & six age-match controls. Vergence eye movements were made to follow targets placed at 20 and 240 cm along the mid-sagittal plane. Eye movements were measured with high-resolution video oculography.
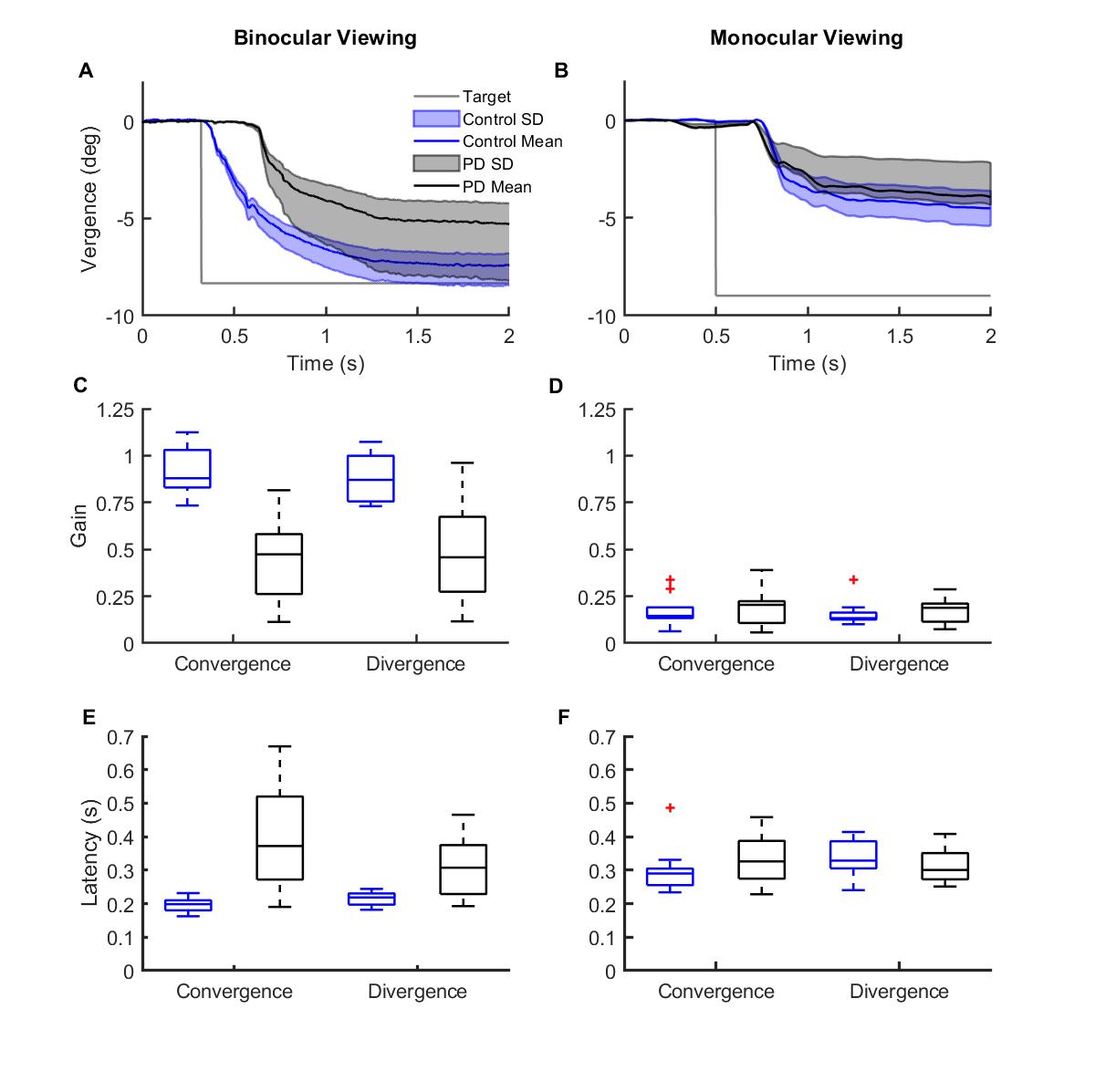
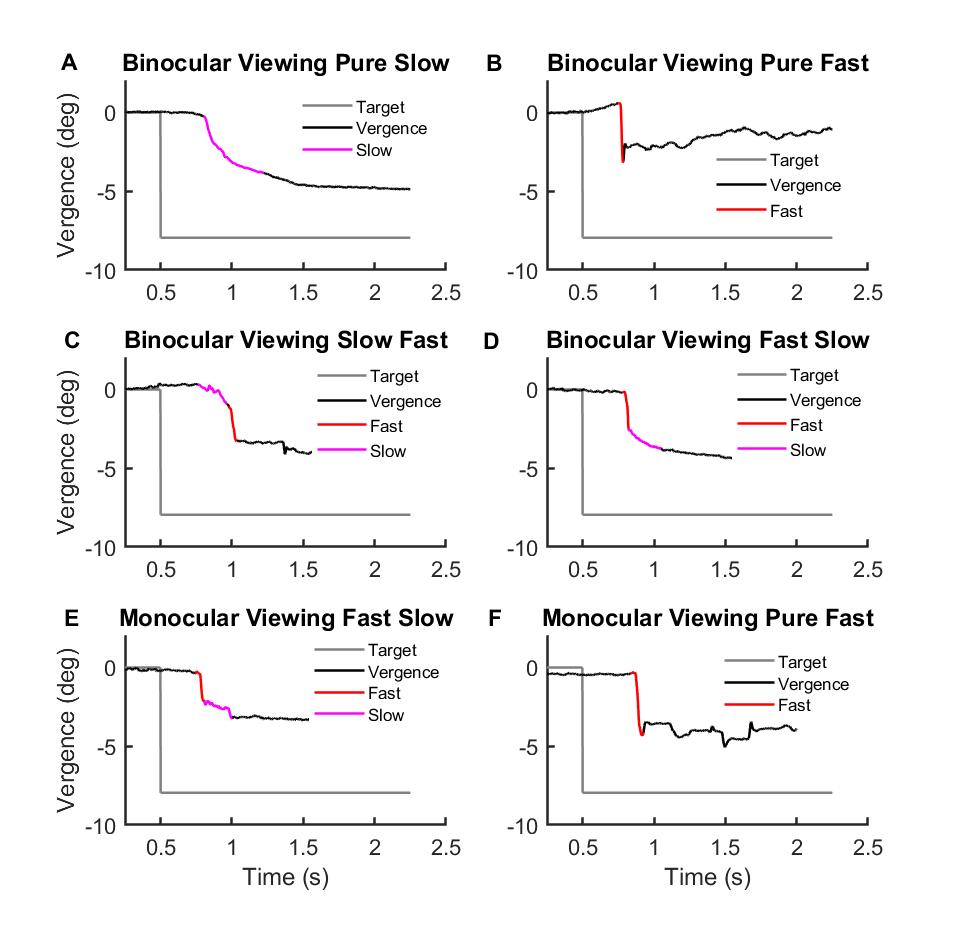
Results: We found differences between BD & DD vergence. Gain (actual eye amplitude/target amplitude) was reduced in PD for DD vergence but not for BD vergence (vs. controls) (Fig 2A-D, PDD < 0.001, PBD =0.66). The latency of DD vergence onset was significantly longer for PD patients compared to controls (Fig 2 E-F, PDD < 0.001, PBD >0.05). Four strategies compensated for the impaired DD vergence; pure slow vergence, pure fast saccadic movements, slow followed by fast movements & fast followed by slow movements (Fig 3 A-D).. BD vergence had only two strategies, fast followed by slow or pure fast saccadic movements (Fig 3 E-F).
[figure 2]
[figure 3]
Conclusion: We found that DD vergence is affected compared to healthy controls, but BD is comparable to healthy subjects. The results suggest involvement of disparity controller in Fig 1. Anatomical connectivity patterns speculate that the deep cerebellar nuclei are disparity controllers. PD affects the cerebellar outflow via the subthalamic connections to the cerebellum.
References: 1. Fincham, E.F. and J. Walton, The reciprocal actions of accommodation and convergence. J Physiol, 1957. 137(3): p. 488-508. 2. Hung, G.K. and J.L. Semmlow, Static behavior of accommodation and vergence: computer simulation of an interactive dual-feedback system. IEEE Trans Biomed Eng, 1980. 27(8): p. 439-47. 3. Krishnan, V.V., S. Phillips, and L. Stark, Frequency analysis of accommodation, accommodative vergence and disparity vergence. Vision Res, 1973. 13(8): p. 1545-54. 4. Krishnan, V.V. and L. Stark, A heuristic model for the human vergence eye movement system. IEEE Trans Biomed Eng, 1977. 24(1): p. 44-9. 5. Westheimer, G., Amphetamine, Barbiturates, and Accommodation-Convergence. Arch Ophthalmol, 1963. 70: p. 830-6.
To cite this abstract in AMA style:
P. Gupta, S. Beylergil, J. Murray, A. Shaikh, F. Ghasia. Effects of Parkinson’s disease on blur-driven and disparity-driven vergence eye movements [abstract]. Mov Disord. 2021; 36 (suppl 1). https://www.mdsabstracts.org/abstract/effects-of-parkinsons-disease-on-blur-driven-and-disparity-driven-vergence-eye-movements/. Accessed May 21, 2026.« Back to MDS Virtual Congress 2021
MDS Abstracts - https://www.mdsabstracts.org/abstract/effects-of-parkinsons-disease-on-blur-driven-and-disparity-driven-vergence-eye-movements/