Category: Parkinson's Disease: Non-Motor Symptoms
Objective: Determine the viability of a wearable reduced montage EEG device to record sleep in a population of patients with Parkinson disease (PD).
Background: Sleep disorders are among the most common non-motor PD symptoms[1], but the study of these disorders is hampered by a lack of easily obtainable objective data[2,3]. Polysomnography (PSG) is the gold standard for studying and diagnosing sleep disorders. However, PSGs have several limitations: they are expensive, require analysis by trained sleep experts, typically occur in a sleep clinic, and on average, are limited to one night of recording, meaning that they do not capture variability across nights. Recently, wearable devices capable of PSG-validated sleep monitoring have been developed for in-home use[4]. Although they have been validated and studied extensively in young, healthy subjects, no data exists on the utility of these devices in patients with PD.
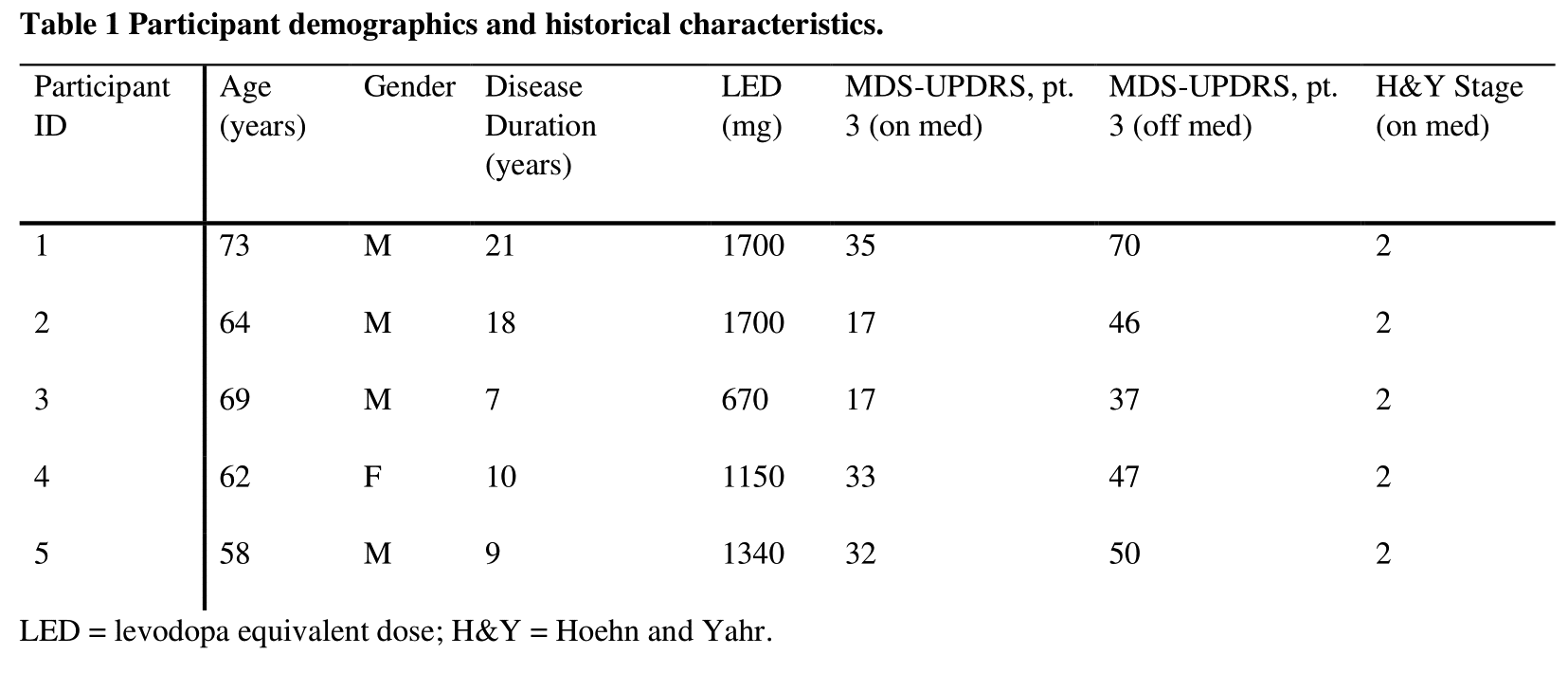
Method: We recorded in-home sleep using a headband device containing 5 dry-EEG electrodes, a 3D accelerometer, and a pulse oximeter for an average of 5 ± 2.2 nights in a group of 5 PD patients (Table 1). Patients continued their usual antiparkinsonian medications, except for one night of medication withdrawal. Parameters measured included total sleep time (TST), sleep efficiency (SE), sleep onset latency (SOL), wake after sleep onset (WASO), and time spent in stages N1, N2, N3, and REM.
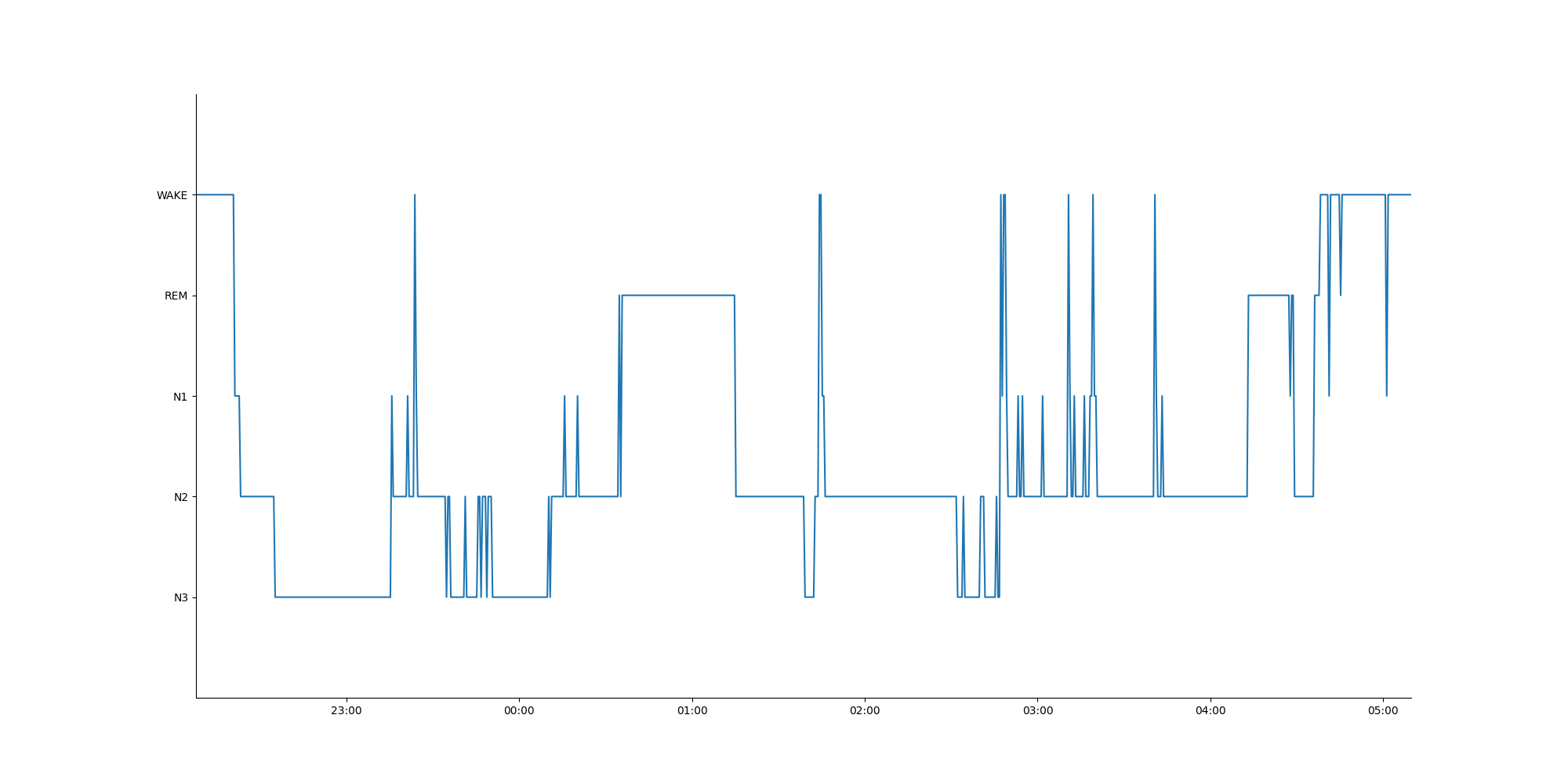
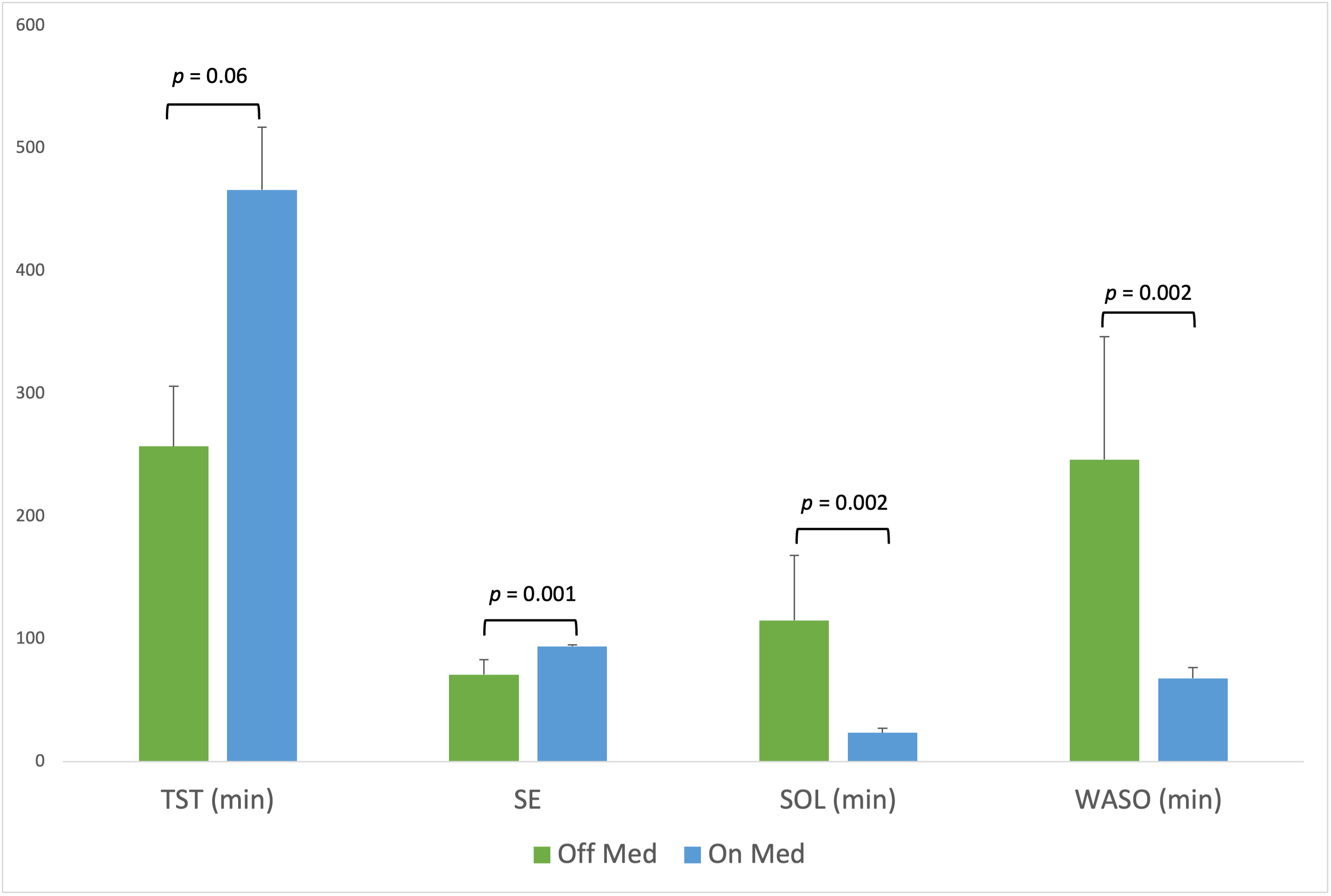
Results: Data was successfully obtained from all 5 subjects. A representative hypnogram from a single night is shown in Figure 2. Group averages (95% CI) for sleep parameters, as determined by the device’s automated sleep staging algorithm, were: TST (min): 424.3 (336.2 – 512.3); SE: 89.5 (83.4 – 95.5); SOL (min): 41.8 (17.2 – 66.5); WASO (min): 103.6 (55.7 – 151.5); N1 (min): 44.8 (32.4 – 57.2); N2 (min): 243.3 (163.2 – 323.4); N3 (min): 72.2 (40.2 – 104.2); and REM (min): 54.4 (27.7 – 81.0). On vs. off medications, subjects had significantly higher SE (on 94.6, off 59.2, p = 0.001), and lower SOL (on 22.2, off 95.9, p = 0.002) and WASO (on 68.1, off 205.2, p = 0.002) (Figure 2). TST was not significantly different on vs. off medication.
Conclusion: Wearable devices can be used to record sleep in PD patients in the home setting. Sleep architecture is worsened during antiparkinsonian medication withdrawal. Future studies will be needed to validate these devices against PSG in this patient population.
References: [1] Barone P, Antonini A, Colosimo C, Marconi R, Morgante L, Avarello TP, et al. The PRIAMO study: A multicenter assessment of nonmotor symptoms and their impact on quality of life in Parkinson’s disease. Mov Disord 2009;24:1641–9. https://doi.org/10.1002/mds.22643.
[2] Chahine LM, Amara AW, Videnovic A. A systematic review of the literature on disorders of sleep and wakefulness in Parkinson’s disease from 2005 to 2015. Sleep Med Rev 2017;35:33–50. https://doi.org/10.1016/j.smrv.2016.08.001.
[3] Baumgartner AJ, Kushida CA, Summers MO, Kern DS, Abosch A, Thompson JA. Basal ganglia local field potentials as a potential biomarker for sleep disturbance in Parkinson’s disease. Front Neurol 2021;12:765203. https://doi.org/10.3389/fneur.2021.765203.
[4] Arnal PJ, Thorey V, Debellemaniere E, Ballard ME, Bou Hernandez A, Guillot A, et al. The Dreem Headband compared to polysomnography for electroencephalographic signal acquisition and sleep staging. Sleep 2020;43:1–13. https://doi.org/10.1093/sleep/zsaa097.
To cite this abstract in AMA style:
J. Thompson, L. Hirt, D. Kern, A. Baumgartner. Characterization of at-home sleep macrostructure in Parkinson disease using a wearable sleep monitoring device [abstract]. Mov Disord. 2023; 38 (suppl 1). https://www.mdsabstracts.org/abstract/characterization-of-at-home-sleep-macrostructure-in-parkinson-disease-using-a-wearable-sleep-monitoring-device/. Accessed May 4, 2026.« Back to 2023 International Congress
MDS Abstracts - https://www.mdsabstracts.org/abstract/characterization-of-at-home-sleep-macrostructure-in-parkinson-disease-using-a-wearable-sleep-monitoring-device/