Category: Parkinsonism, Atypical: PSP, CBD
Objective: To investigate the language impairment profile of patients diagnosed as probable Corticobasal Syndrome (CBS) [1] in light of a multimodal imaging biomarkers analysis, namely brain atrophy patterns, functional and disease-specific imaging biomarkers.
Background: CBS is a neurodegenerative disease related to multiple underlying pathologies [2]. Besides movement disorders and cognitive deficits, it is recognized that language and speech are also commonly impaired [1]. Previous studies described a phenotype similar to the non-fluent variant of primary progressive aphasia [3], whereas others have reported a wide variety of phenotypes [4,5].
Method: Thirty-one patients with probable CBS were prospectively enrolled. A comprehensive language/speech evaluation was performed. They underwent [18F]fluorodeoxyglucose-positron emission tomography (FDG-PET) and [11C]Pittsburgh Compound-B (PIB-PET) on a PET-MRI to assess their amyloid status. Brain atrophy patterns were analyzed using voxel-based morphometry (VBM), and FDG-PET quantitative group analyses were performed at SPM8. A healthy control-group of 30 subjects, matched by age and education, was also included.
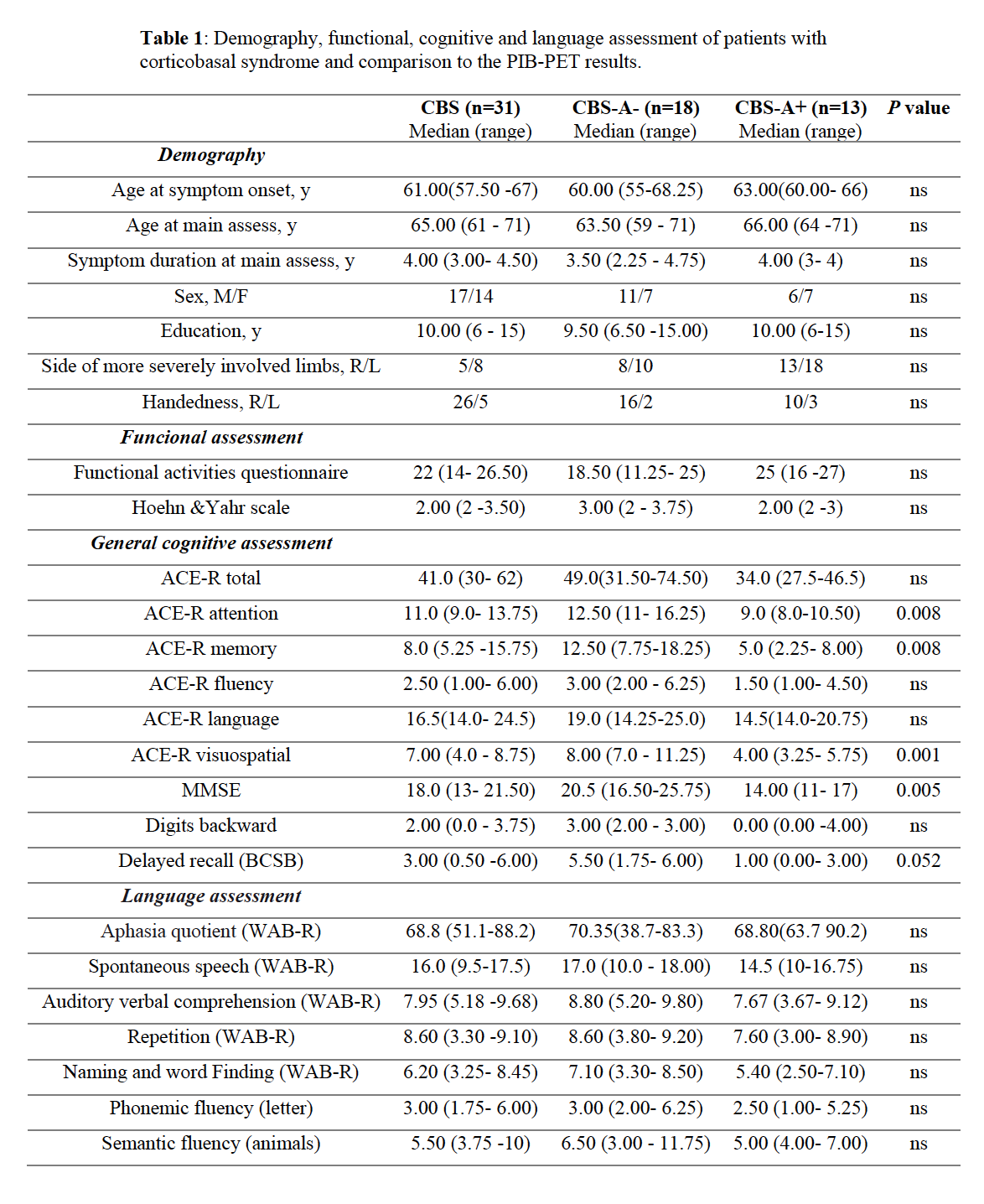
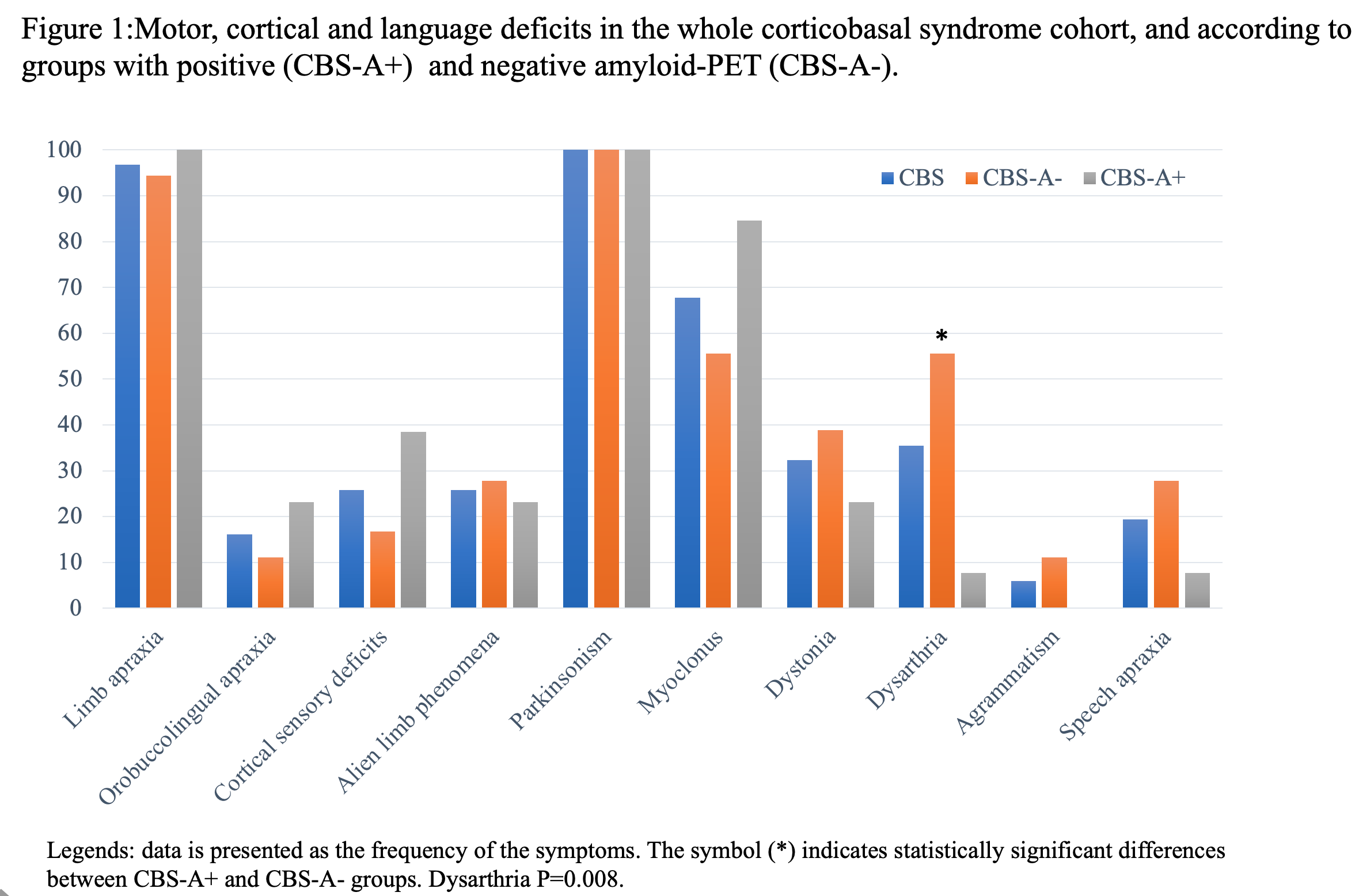
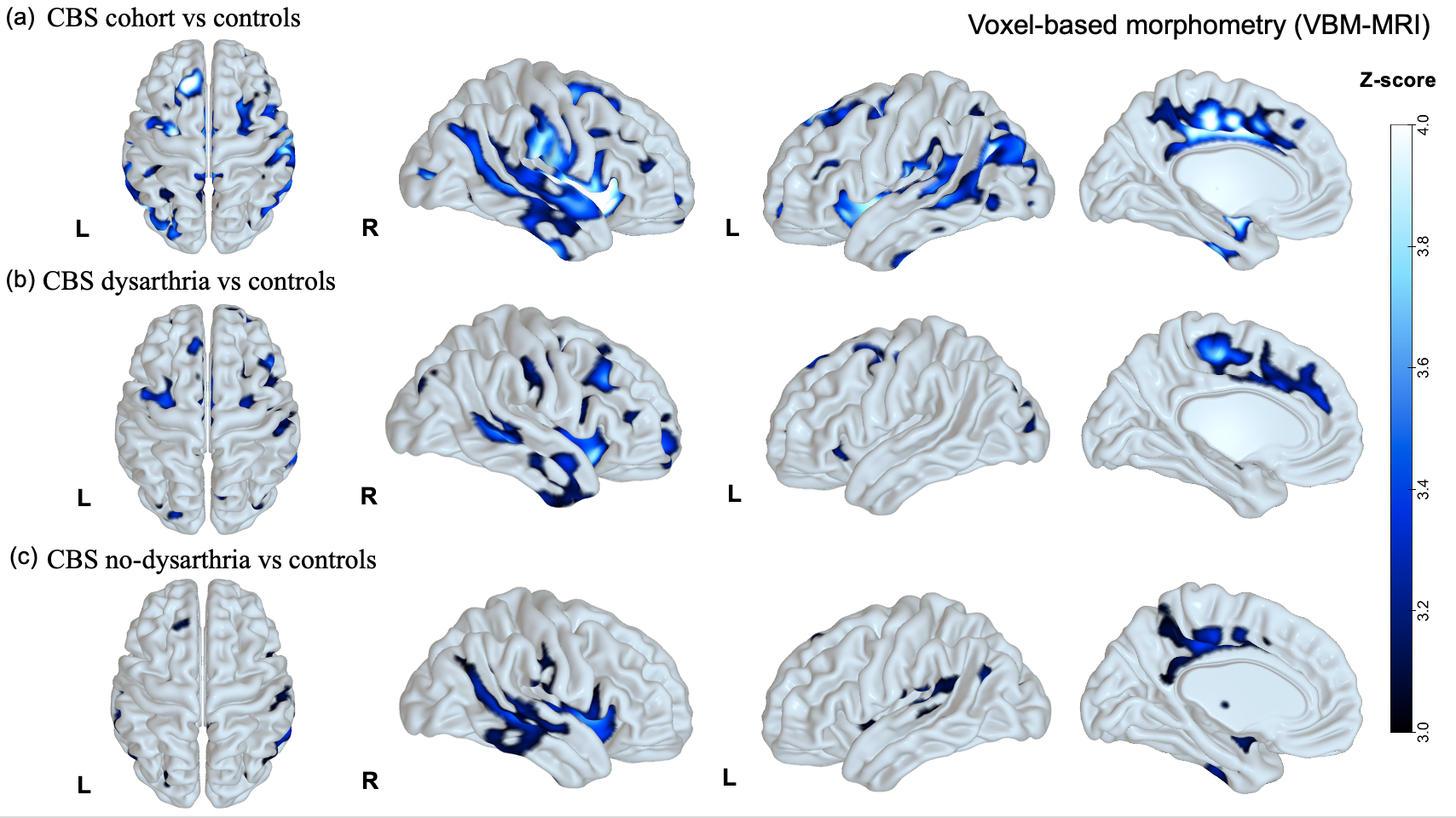
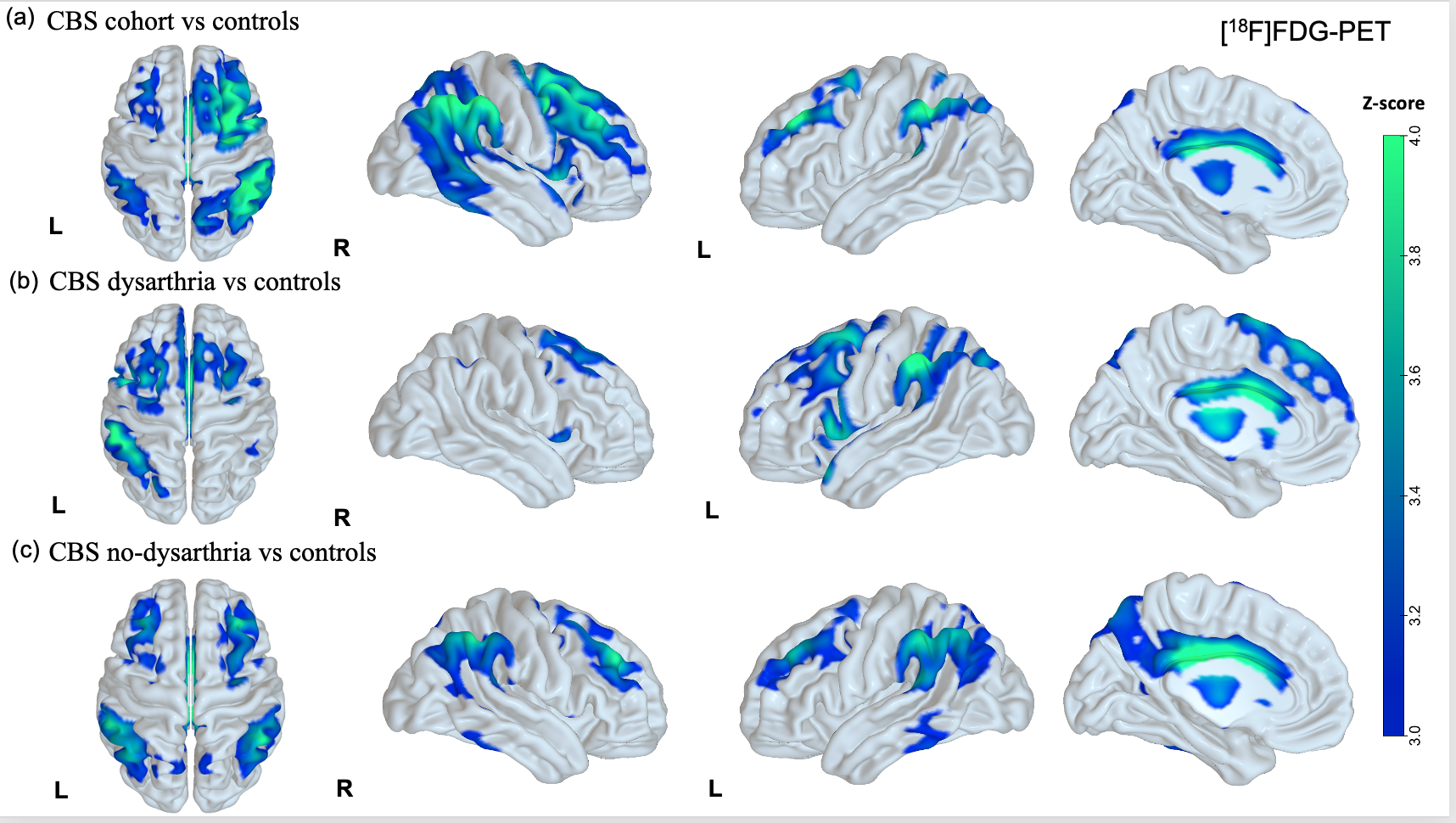
Results: CBS patients with amyloid-positive (CBS-A+,13) and negative imaging (CBS-A-,18) did not differ significantly in demography (Table 1). CBS-A+ group demonstrated a worse performance in the general cognitive assessment, mostly in attention, memory, and visuospatial domains (table 1). Both groups did not differ concerning the language cognitive tests, such as the Western Battery aphasia-Revised and verbal fluency tasks. Noteworthy, CBS-A- patients displayed dysarthria more often than CBS-A+ (p=0.008) (figure 1). CBS patients had frontoparietal and basal ganglia hypometabolic patterns compared to controls (figure 2 (a)). Patients with dysarthria showed hypometabolism in the left inferior and middle frontal gyrus, and left thalamus and caudate (figure 2 (b)). They also demonstrated brain atrophy at bilateral caudate and left supplementary motor area (SMA) (figure 3 (b)).
Conclusion: Language impairment in CBS is highly heterogeneous. Dysarthria may be a valuable clinical clue suggesting underlying pathology, probably more related to CBS due to tauopathies than Alzheimer’s disease. Left frontal areas, the SMA, and basal ganglia might play a role in manifesting speech disorders related to tauopathies.
References: [1] Armstrong MJ, Litvan I, Lang AE, et al. Criteria for the diagnosis of corticobasal degeneration. Neurology 2013; 80: 496–503. [2] Boeve BF, Lang AE, Litvan I. Corticobasal degeneration and its relationship to progressive supranuclear palsy and frontotemporal dementia. Ann Neurol 2003; 54: 15–9. [3] Shelley BP, Hodges JR, Kipps CM, Xuereb JH, Bak TH. Is the pathology of corticobasal syndrome predictable in life? Mov Disord 2009; 24: 1593–9. [4] Peterson KA, Patterson K, Rowe JB. Language impairment in progressive supranuclear palsy and corticobasal syndrome. J Neurol 2019. [5] Burrell JR, Hornberger M, Villemagne VL, Rowe CC, Hodges JR. Clinical Profile of PiB-Positive Corticobasal Syndrome. PLoS One 2013; 8.
To cite this abstract in AMA style:
J. Parmera, M. Oliveira, A. Coutinho, I. de Almeida, C. Carneiro, A. Neto, R. Nitrini, C. Buchpiguel, E. Barbosa, S. Brucki. Language impairment in Corticobasal Syndrome: from clinical phenotype to biomarkers [abstract]. Mov Disord. 2021; 36 (suppl 1). https://www.mdsabstracts.org/abstract/language-impairment-in-corticobasal-syndrome-from-clinical-phenotype-to-biomarkers/. Accessed April 28, 2026.« Back to MDS Virtual Congress 2021
MDS Abstracts - https://www.mdsabstracts.org/abstract/language-impairment-in-corticobasal-syndrome-from-clinical-phenotype-to-biomarkers/