Session Information
Date: Wednesday, September 25, 2019
Session Title: Non-Motor Symptoms
Session Time: 1:15pm-2:45pm
Location: Agora 3 West, Level 3
Objective: To evaluate cognitive correlates of nocturnal sleep dysfunction in patients with idiopathic Parkinson’s disease (PD).
Background: Sleep disturbances are among the most troublesome and frequent non-motor symptoms of PD which been associated with cognitive impairment[1, 2]; nevertheless, research has suggested that nighttime sleep problems are not related to cognitive status [3].
Method: Retrospective analytical study. Case files of PD patients were reviewed, and those with complete Parkinson’s Disease Sleep Scale (PDSS) and Montreal Cognitive Assessment (MoCA), Hoehn & Yahr and MDS-UPDRS, were included for analysis. Spearman’s correlations were determined between PDSS total and subscores and cognitive (MoCA) scores.
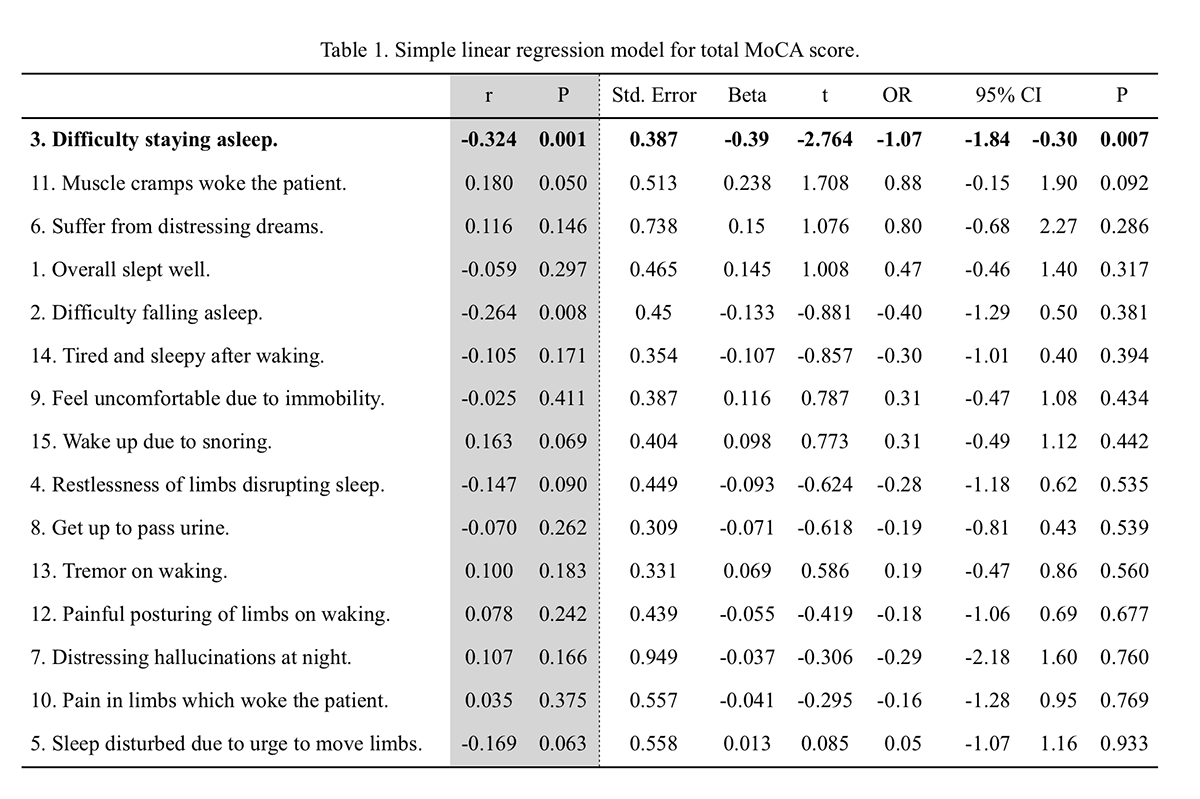
Results: A total sample of 84 patients was analyzed, 57 (68%) male, mean age 60.4±11.6, age at onset 55.2±12.3 and disease duration 6.7±5.5 years. Mean Hoehn & Yahr 2.3±0.8, and MDS-UPDRS III 42.2±24.5. Mean PDSS score was 15.3±10.1; the most frequent disturbance was nocturia (n=61, 73%), followed by inability to move (n=46, 55%), and morning tiredness (n=45, 57%). Mean MoCA 24.0±4.2, 13 (16%) patients had a score <21, with the most impaired domains: delayed recall (mean 2.2 points, 44% of total) and visuospatial domain (mean 3.6 points, 72% of total). Significant correlations were between the total PDSS score and patient-reported experiences of daily life, in both the non-motor (r=0.59, p<0.01) and motor (r=0.49, p<0.01) domains, along with disease severity (r=0.26, p<0.05), duration (r=0.25, p<0.05), and motor complications of treatment (r=0.37, p<0.01). Among PDSS subscores, those related to insomnia (difficulty falling and staying asleep) were significantly correlated to the MoCA score (r=-0.26, p=0.008, and r= -0.32, p=0.001 respectively). A linear regression model (Table 1) with MoCA total score as dependent variable and PDSS subscores as predictive variables, revealed that only late insomnia (difficulty staying asleep) was a significant predictor of cognitive status (OR -1.70, 95%CI -1.84– -0.30; p=0.007).
Conclusion: In patients with Parkinson’s disease, insomnia is predictive of cognitive status; although our sample is small, significant correlations between nocturnal sleep dysfunction and disease stage, severity, and duration, are in concordance with results of previous studies, adding soundness to our results.
References: 1. Goldman JG, Stebbins GT, Leung V, Tilley BC, Goetz CG. Relationships among cognitive impairment, sleep, and fatigue in Parkinson’s disease using the MDS-UPDRS. Parkinsonism Relat Disord. 2014;20(11):1135-9. 2. Pushpanathan ME, Loftus AM, Thomas MG, Gasson N, Bucks RS. The relationship between sleep and cognition in Parkinson’s disease: A meta-analysis. Sleep Med Rev. 2016;26:21-32. 3. Goldman JG, Ghode RA, Ouyang B, Bernard B, Goetz CG, Stebbins GT. Dissociations among daytime sleepiness, nighttime sleep, and cognitive status in Parkinson’s disease. Parkinsonism Relat Disord. 2013;19(9):806-11.
To cite this abstract in AMA style:
S. Castillo-Torres, C. Cerda-Contreras, D. Roque-Martínez, D. Díaz-Pérez, X. Ortiz-Jiménez, B. Chávez-Luévanos, I. Estrada-Bellmann. (Not) sleeping with the enemy: Cognitive correlates of nocturnal sleep disturbances in Parkinson’s disease [abstract]. Mov Disord. 2019; 34 (suppl 2). https://www.mdsabstracts.org/abstract/not-sleeping-with-the-enemy-cognitive-correlates-of-nocturnal-sleep-disturbances-in-parkinsons-disease/. Accessed May 8, 2026.« Back to 2019 International Congress
MDS Abstracts - https://www.mdsabstracts.org/abstract/not-sleeping-with-the-enemy-cognitive-correlates-of-nocturnal-sleep-disturbances-in-parkinsons-disease/