Objective: To evaluate the long-term effectiveness of CLES on FOG and treatment satisfaction in APD patients.
Background: FOG, prevalent in about 60% of APD patients, worsens with disease progression and is associated with falls, dyskinesia, decreased mobility, and decreased quality of life. Worsening of motor symptoms in APD patients is associated with dissatisfaction with treatment. Since CLES is device aided therapy, it is important to explore not only the effectiveness of CLES on symptoms such as FOG, but also the treatment satisfaction. Currently, there is limited understanding of the long-term impact of CLES on FOG and treatment satisfaction.
Method: PROviDE is a 3-year prospective, home-based, observational study examining the effectiveness of CLES treatment on patient-reported outcomes [1]. The primary objectives of this interim analysis were to evaluate the change in FOG and treatment satisfaction at 12 months after CLES initiation. FOG was measured using FOGQ-sa, the self-administered version of FOGQ [2]. Treatment satisfaction was measured using TSQM-9 which assesses patient’s satisfaction with medication on effectiveness, convenience, and global satisfaction [3]. Change in off time was also evaluated using modified UPDRS Part IV Item 39 at 12 months.
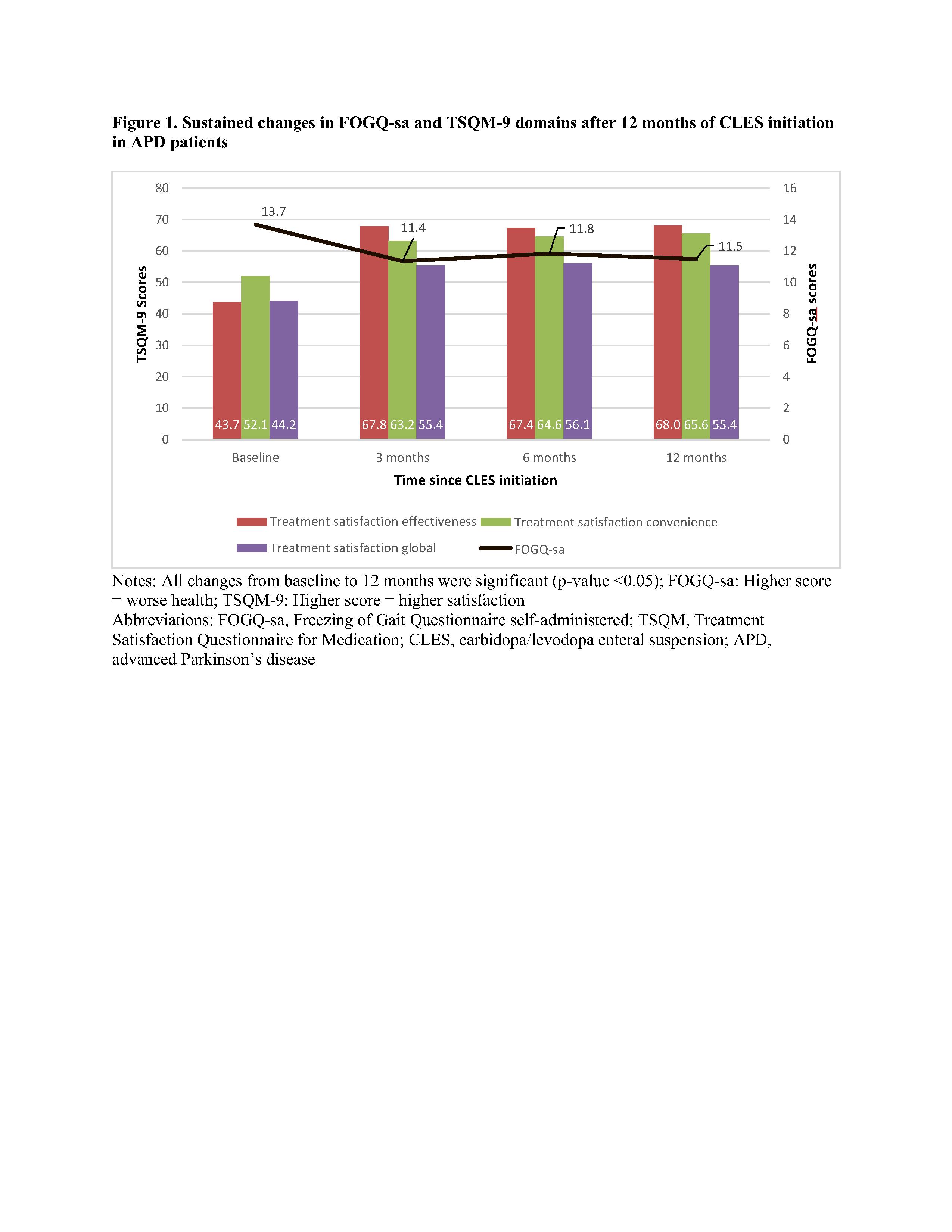
Results: In our sample (N=83), the mean FOGQ-sa score reduced significantly at 12 months after CLES initiation by 1.83 (95% CI: -2.86 – -0.80; p-value <0.05) [Figure 1]. At 12 months, significant improvements (p-value <0.0001) were also observed in treatment satisfaction score for effectiveness (mean: 24.33, 95% CI: 20.36 – 28.29), convenience (mean: 13.51; 95% CI: 9.08 – 17.95), and global satisfaction (mean: 10.89; 95% CI: 6.71 – 15.07). These patients also demonstrated significant reduction in off-time (mean: -2.84; 95% CI: -4.25 – -1.43; p-value <0.0001). These significant improvements were seen at 3 months [4] and were sustained through 12 months.
Conclusion: This study demonstrates real-world sustained effectiveness of CLES in reducing FOG, off time, and improving treatment satisfaction in one of the largest US-specific cohorts of APD patients with CLES. Despite being a device aided therapy, CLES is a valuable treatment option for APD with sustained patient satisfaction with treatment.
References: 1. Pahwa R. Evaluating long-term effectiveness of carbidopa/levodopa enteral suspension in advanced Parkinson’s Disease patients: PROviDE study design and baseline characteristics. Movement disorders. 2018;33:S93. 2. Nilsson, M. H., Hariz, G. M., Wictorin, K., Miller, M., Forsgren, L., & Hagell, P. (2010). Development and testing of a self administered version of the Freezing of Gait Questionnaire. BMC neurology, 10(1), 85. 3. Bharmal, M., Payne, K., Atkinson, M. J., Desrosiers, M. P., Morisky, D. E., & Gemmen, E. (2009). Validation of an abbreviated Treatment Satisfaction Questionnaire for Medication (TSQM-9) among patients on antihypertensive medications. Health and quality of life outcomes, 7(1), 36. 4. Pahwa, R., Isaacson, S., Kandukuri, P., Jalundhwala, Y., Kukreja, P., Bao, Y., … & Aldred, J. (2019). Evaluating the real-world effectiveness of carbidopa/levodopa enteral suspension (CLES) on patients with advanced Parkinson’s disease in USA: Interim-evidence from the PROviDE study: 169. Movement Disorders, 34.
To cite this abstract in AMA style:
R. Pahwa, S. Isaacson, B. Bluett, P. Kandukuri, N. Gupta, Y. Jalundhwala, P. Kukreja, I. Pan, J. Aldred. Sustained Improvement in Freezing of Gait (FOG) and Treatment Satisfaction at 12 months After Initiation of Carbidopa/Levodopa Enteral Suspension (CLES) in Advanced Parkinson’s Disease (APD) Patients in USA: Interim Results from the PROviDE Study [abstract]. Mov Disord. 2020; 35 (suppl 1). https://www.mdsabstracts.org/abstract/sustained-improvement-in-freezing-of-gait-fog-and-treatment-satisfaction-at-12-months-after-initiation-of-carbidopa-levodopa-enteral-suspension-cles-in-advanced-parkinsons-disease-apd-p/. Accessed May 21, 2026.« Back to MDS Virtual Congress 2020
MDS Abstracts - https://www.mdsabstracts.org/abstract/sustained-improvement-in-freezing-of-gait-fog-and-treatment-satisfaction-at-12-months-after-initiation-of-carbidopa-levodopa-enteral-suspension-cles-in-advanced-parkinsons-disease-apd-p/