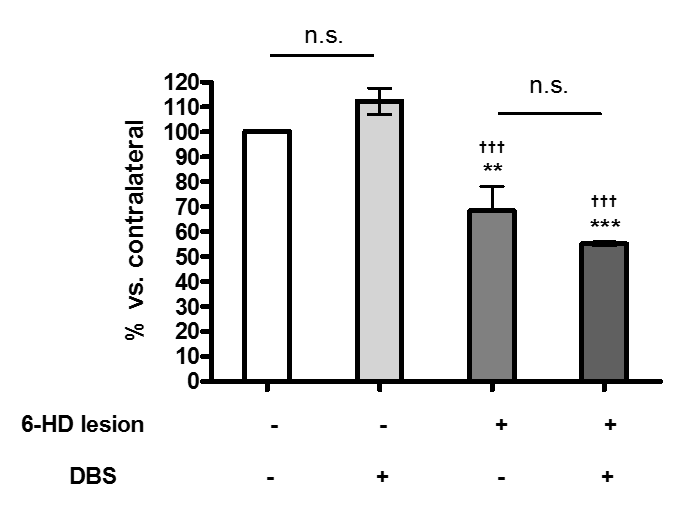Session Information
Date: Wednesday, September 25, 2019
Session Title: Surgical Therapy
Session Time: 1:15pm-2:45pm
Location: Les Muses Terrace, Level 3
Objective: Globus pallidus interna (GPi) is recognized as an effective treatment for advanced Parkinson’s disease. However, the neurochemical basis of its effects remains unknown. The aim of this study was to investigate effects of entopeduncular nucleus (EPN) stimulation, the equivalent to human GPi, in sham and 6-hydroxydopamine (6-OHDA)-lesioned rat model on changes of glutamate and GABA in the striatum.
Background: Parkinson’s disease (PD) is a neurodegenerative disorder characterized by dopamine depletion due to the degeneration of substantia nigral dopamine neurons. Globus pallidus interna (GPi) stimulation has become an established treatment for motor symptoms of PD. However, the exact mechanism of deep brain stimulation (DBS) in GPi stimulation is unknown.
Method: Extracellular glutamate and GABA level changes in striatum of sham group, sham plus deep brain stimulation (DBS) group, 6-OHDA group, and 6-OHDA plus DBS group were examined using microdialysis and high-pressure liquid chromatography (HPLC). Tyrosine hydroxylase (TH) immunoreactivities in substantia nigra and striatum of the four groups were also analyzed.
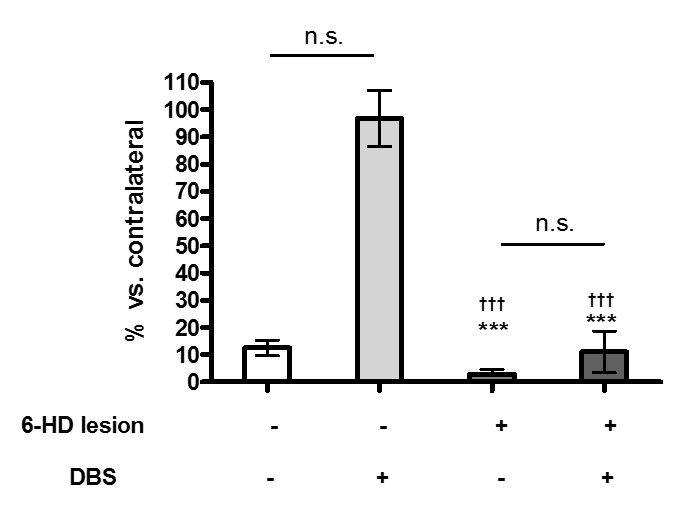
Results: Extracellular glutamate levels in the striatum of sham plus DBS group and 6-OHDA plus DBS group were significant increased by EPN stimulation compared to those in the sham group and 6-OHDA group. However, extracellular GABA levels of the striatum of sham plus DBS group and 6-OHDA plus DBS group had no significant changes by EPN stimulation compared to those in the sham group and 6-OHDA group. EPN stimulation had no significant effect on the expression of tyrosine hydroxylase in sham or 6-OHDA group.
Conclusion: Our results provided new neurochemical data of EPN stimulation. Clinical results of EPN stimulation are not restricted to direct inhibitory outflow to thalamus. They also involve widespread adaptive changes within the basal ganglia.
References: 1. Agnesi F, Blaha CD, Lin J, Lee KH: Local glutamate release in the rat ventral lateral thalamus evoked by high-frequency stimulation. J Neural Eng 7: 26009, 2010. 2. Bar Gad I, Elias S, Vaadia E, Bergman H: Complex locking rather than complete cessation of neuronal activity in the globus pallidus of a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-treated primate in response to pallidal microstimulation. The Journal of neuroscience 24: 7410-7419, 2004. 3. Barneoud P, Parmentier S, Mazadier M, Miquet JM, Boireau A, Dubedat P, et al.: Effects of complete and partial lesions of the dopaminergic mesotelencephalic system on skilled forelimb use in the rat. Neuroscience 67: 837-848, 1995. 4. Boulet S, Lacombe E, Carcenac C, Feuerstein C, Sgambato-Faure V, Poupard A, et al.: Subthalamic stimulation-induced forelimb dyskinesias are linked to an increase in glutamate levels in the substantia nigra pars reticulata. J Neurosci 26: 10768-10776, 2006. 5. Bruet N, Windels F, Carcenac C, Feuerstein C, Bertrand A, Poupard A, et al.: Neurochemical mechanisms induced by high frequency stimulation of the subthalamic nucleus: increase of extracellular striatal glutamate and GABA in normal and hemiparkinsonian rats. Journal of Neuropathology and Experimental Neurology 62: 1228-1240, 2003. 6. DeLong MR: Primate models of movement disorders of basal ganglia origin. Trends in neurosciences 13: 281-285, 1990. 7. DeLong MR, Wichmann T: Basal Ganglia Circuits as Targets for Neuromodulation in Parkinson Disease. JAMA Neurol 72: 1354-1360, 2015. 8. Gubellini P, Eusebio A, Oueslati A, Melon C, Kerkerian-Le Goff L, Salin P: Chronic high-frequency stimulation of the subthalamic nucleus and L-DOPA treatment in experimental parkinsonism: effects on motor behaviour and striatal glutamate transmission. Eur J Neurosci 24: 1802-1814, 2006. 9. Hazrati LN, Parent A: Differential patterns of arborization of striatal and subthalamic fibers in the two pallidal segments in primates. Brain research 598: 311-315, 1992. 10. Jonkers N, Sarre S, Ebinger G, Michotte Y: MK801 suppresses the L-DOPA-induced increase of glutamate in striatum of hemi-Parkinson rats. Brain Res 926: 149-155, 2002. 11. Lanciego JL, Gonzalo N, Castle M, Sanchez-Escobar C, Aymerich MS, Obeso JA: Thalamic innervation of striatal and subthalamic neurons projecting to the rat entopeduncular nucleus. Eur J Neurosci 19: 1267-1277, 2004. 12. Lang AE, Widner H: Deep brain stimulation for Parkinson’s disease: patient selection and evaluation. Mov Disord 17 Suppl 3: S94-101, 2002. 13. Lee KH, Kristic K, van Hoff R, Hitti FL, Blaha C, Harris B, et al.: High-frequency stimulation of the subthalamic nucleus increases glutamate in the subthalamic nucleus of rats as demonstrated by in vivo enzyme-linked glutamate sensor. Brain Res 1162: 121-129, 2007. 14. Lee KJ, Shim I, Sung JH, Hong JT, Kim IS, Cho CB: Striatal Glutamate and GABA after High Frequency Subthalamic Stimulation in Parkinsonian Rat. J Korean Neurosurg Soc 60: 138-145, 2017. 15. Liu Y, Li W, Tan C, Liu X, Wang X, Gui Y, et al.: Meta-analysis comparing deep brain stimulation of the globus pallidus and subthalamic nucleus to treat advanced Parkinson disease. J Neurosurg 121: 709-718, 2014. 16. McConnell GC, So RQ, Hilliard JD, Lopomo P, Grill WM: Effective deep brain stimulation suppresses low-frequency network oscillations in the basal ganglia by regularizing neural firing patterns. J Neurosci 32: 15657-15668, 2012. 17. McIntyre CC, Savasta M, Kerkerian-Le Goff L, Vitek JL: Uncovering the mechanism(s) of action of deep brain stimulation: activation, inhibition, or both. Clin Neurophysiol 115: 1239-1248, 2004. 18. Odekerken VJJ, van Laar T, Staal MJ, Mosch A, Hoffmann CFE, Nijssen PCG, et al.: Subthalamic nucleus versus globus pallidus bilateral deep brain stimulation for advanced Parkinson’s disease (NSTAPS study): a randomised controlled trial. The Lancet Neurology 12: 37-44, 2013. 19. Papa SM, Engber TM, Kask AM, Chase TN: Motor fluctuations in levodopa treated parkinsonian rats: relation to lesion extent and treatment duration. Brain research 662: 69-74, 1994. 20. Paxinos G: The rat nervous system. Amsterdam ; Boston: Elsevier Academic Press, 2004. 21. Shipton EA: Movement disorders and neuromodulation. Neurol Res Int 2012: 309431, 2012. 22. Tan ZG, Zhou Q, Huang T, Jiang Y: Efficacies of globus pallidus stimulation and subthalamic nucleus stimulation for advanced Parkinson’s disease: a meta-analysis of randomized controlled trials. Clin Interv Aging 11: 777-786, 2016. 23. Temel Y, Visser-Vandewalle V, Aendekerk B, Rutten B, Tan S, Scholtissen B, et al.: Acute and separate modulation of motor and cognitive performance in parkinsonian rats by bilateral stimulation of the subthalamic nucleus. Exp Neurol 193: 43-52, 2005. 24. Wichmann T, Delong M: Deep-Brain Stimulation for Basal Ganglia Disorders. Basal Ganglia 1: 65-77, 2011.
To cite this abstract in AMA style:
CB. Cho, JH. Sung. Change of extracellular glutamate and GABA level in striatum during deep brain stimulation of the entopeduncular nucleus in rats [abstract]. Mov Disord. 2019; 34 (suppl 2). https://www.mdsabstracts.org/abstract/change-of-extracellular-glutamate-and-gaba-level-in-striatum-during-deep-brain-stimulation-of-the-entopeduncular-nucleus-in-rats/. Accessed May 4, 2026.« Back to 2019 International Congress
MDS Abstracts - https://www.mdsabstracts.org/abstract/change-of-extracellular-glutamate-and-gaba-level-in-striatum-during-deep-brain-stimulation-of-the-entopeduncular-nucleus-in-rats/