Objective: KINETIC 2 (NCT05173012) is an ongoing, phase 2b, randomized, double-blind, placebo-controlled, dose-response study that aims to evaluate the efficacy, safety, and tolerability of SAGE-324/BIIB124 (SAGE-324) in patients with essential tremor (ET).
Background: Approximately 50% of patients do not respond to standard-of-care treatments for ET [1-3]. Gamma-aminobutyric acid (GABA) dysregulation has been implicated in ET pathophysiology. SAGE-324 is an investigational, positive allosteric modulator of GABA type A receptors. In a prior phase 2 trial (KINETIC), patients with ET who received 60 mg of SAGE-324 once daily in the morning experienced a statistically significant reduction in upper limb tremor at Day 29 (as assessed by change from baseline [CFB] in The Essential Tremor Rating Assessment Scale-Performance Subscale [TETRAS-PS] Item 4) compared with those who received placebo; 97.1% of patients who received SAGE-324 had ≥ 1 treatment-emergent adverse event.
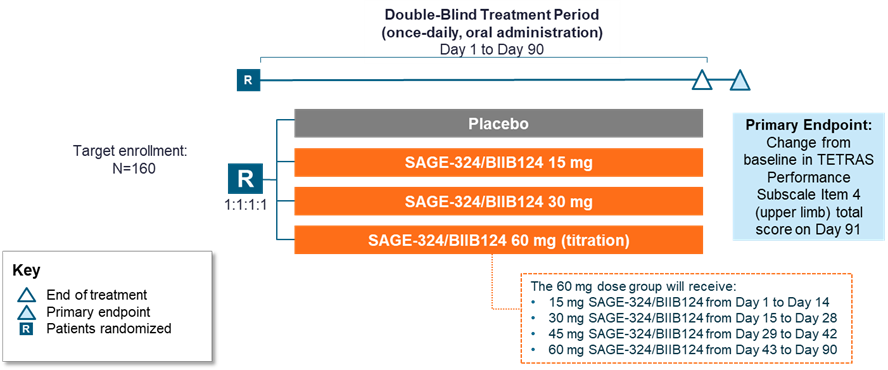
Method: KINETIC 2 aims to enroll approximately 160 patients. Key eligibility criteria include: aged 18–80 years; ET diagnosis (≥ 3 years of isolated tremor syndrome consisting of bilateral upper limb action tremor with or without tremor in other locations); no other relevant neurological signs; TETRAS-PS Item 4 score ≥ 12 and ≥ 6 for the dominant upper limb (total) at screening and predose at Day 1; and a baseline TETRAS Activities of Daily Living (ADL) score ≥ 20 at screening.
Patients are randomized 1:1:1:1 to 4 treatments (15 mg, 30 mg, or 60 mg SAGE-324 [uptitrated from 15 mg over 42 days], or placebo; oral, daily at night for 90 days). [figure1]
The primary endpoint is CFB in TETRAS-PS Item 4 score on Day 91; the secondary endpoint is CFB in TETRAS ADL composite score. Safety and tolerability of SAGE-324 will be evaluated.
Results: KINETIC 2 is estimated to complete in early 2024. Trial updates will be provided at the time of presentation.
Conclusion: KINETIC 2 was designed to evaluate SAGE-324 dose response on clinically relevant endpoints and safety in patients with ET. Enrollment is ongoing, and the results of this trial will inform future SAGE-324 clinical development.
References: [1] Louis ED. Front Neurol. 2012;2:91.
[2] Deuschl G, et al. Lancet Neurol. 2011;10:148-161.
[3] Zesiewicz TA, et al. Neurology. 2011;77:1752-1755.
To cite this abstract in AMA style:
R. Pahwa, A. Ellenbogen, D. Arkilo, T. Dam, T. Lieu, B. Hersh, M. Qin, S. Garafola, M. Gerbasi, H. Colquhoun. KINETIC 2: An ongoing phase 2b, randomized, dose-response study of SAGE-324/BIIB124 for the treatment of essential tremor [abstract]. Mov Disord. 2023; 38 (suppl 1). https://www.mdsabstracts.org/abstract/kinetic-2-an-ongoing-phase-2b-randomized-dose-response-study-of-sage-324-biib124-for-the-treatment-of-essential-tremor/. Accessed July 1, 2025.« Back to 2023 International Congress
MDS Abstracts - https://www.mdsabstracts.org/abstract/kinetic-2-an-ongoing-phase-2b-randomized-dose-response-study-of-sage-324-biib124-for-the-treatment-of-essential-tremor/