Session Information
Date: Monday, September 23, 2019
Session Title: Huntington’s Disease
Session Time: 1:45pm-3:15pm
Location: Agora 3 West, Level 3
Objective: To examine the effects of natural aging for those without Huntington disease (HD) gene expansion and to develop normative curves by age, sex, and education from the distribution of observed scores for the Symbol Digit Modalities Test (SDMT), Stroop Word Reading Test (SWRT), Total Motor Score (TMS), and Total Functional Capacity (TFC) from the UHDRS along with their composite, known as the composite UHDRS (cUHDRS) [1].
Background: The progression of HD for gene-expanded carriers (HDGEC) is well-studied. Natural aging effects, however, are not often considered in the evaluation of HD progression.
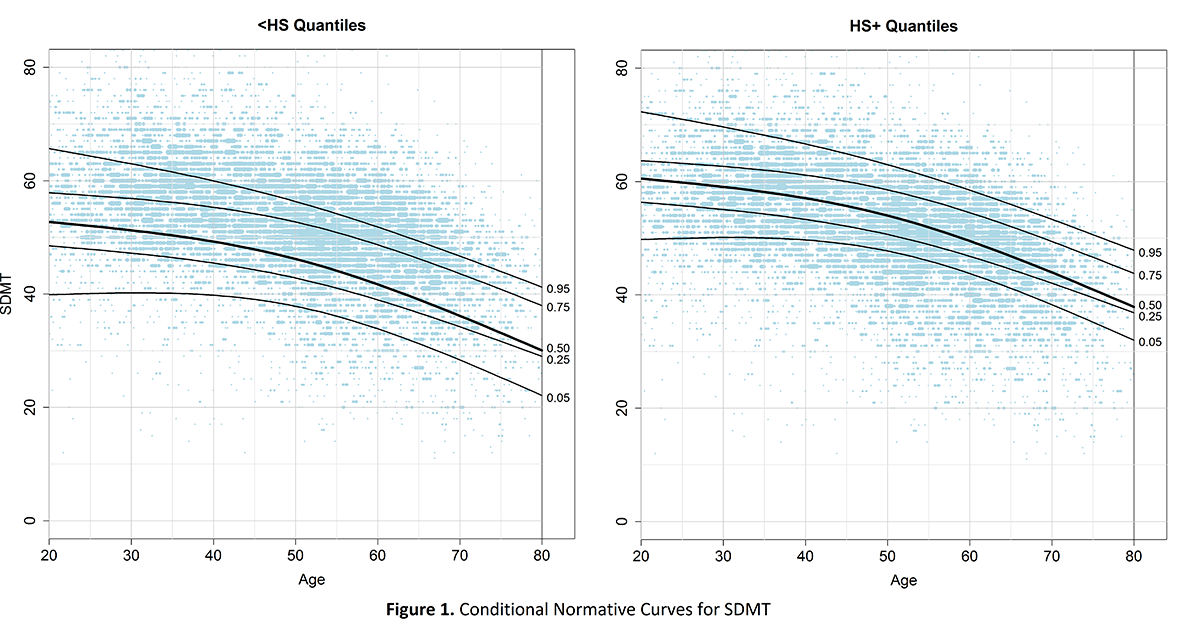
Method: After combining longitudinal REGISTRY [2] and Enroll-HD [3] data from the most recent periodic dataset (PDS4 from Enroll-HD), we fit natural cubic splines with a knot at the median age using linear quantile mixed models [4] for participants without HD gene expansion (N = 3731, N observations = 9451). Normative curves were estimated for the 0.05, 0.25, 0.50, 0.75, and 0.95 quantiles. Two types of reference curves were considered; unconditional curves were only dependent on age while conditional curves were dependent on age and other covariates (e.g., education and/or sex) [5].
Results: Conditioning on education was necessary for SDMT, SWRT, and cUHDRS. Unconditional curves were sufficient for the TMS. TFC was unique in that the curve was constant over age with its intercept being at the maximum score (TFC=13). For all measures, sex effects were minimal so conditioning on sex was unnecessary. [figure1]
Conclusion: Extreme quantile estimates (e.g., the 0.05 quantile) for each measure (SDMT, SWRT, TMS, and cUHDRS) can be considered as boundaries for natural aging and HDGEC scores falling beyond these thresholds are likely the result of HD progression. Normative curves and tables are developed that can serve as references for clinical characterization of HDGEC.
References: 1. Schobel SA, Palermo G, Auinger P, Long JD, et al. Motor, cognitive, and functional declines contribute to a single progressive factor in early HD. Neurology. 2017;89(24):2495-2502. 2. Orth M, Handley OJ, Schwenke C, Landwehrmeyer B. Observing Huntington’s disease: the European Huntington’s disease network’s REGISTRY. Journal of Neurology, Neurosurgery, and Psychiatry. 2010;82:1409–12. 3. Landwehrmeyer BG, Fitter-Attas C, Giuliano J, et al. Data analytics from Enroll-HD, a global clinical research platform for Huntington’s disease. Movement Disorder Clinical Practice. 2016;4:212–24. 4. Geraci M and Bottai M. Linear quantile mixed models. Statistics and Computing. 2014;24(3), 461–479. 5. Wei Y, Pere A, Koenker RW, He X. Quantile regression methods for reference growth charts. Statistics in Medicine. 2006 ;25(8):1369-1382. An earlier version of this abstract was accepted as a poster presentation at the HD Therapeutics Conference, Palm Springs, CA, February 25-28, 2019.
To cite this abstract in AMA style:
J. Mills, J. Long, A. Mohan, J. Ware, C. Sampaio. Normative Curves for Selected Unified Huntington Disease Rating Scale Measures [abstract]. Mov Disord. 2019; 34 (suppl 2). https://www.mdsabstracts.org/abstract/normative-curves-for-selected-unified-huntington-disease-rating-scale-measures/. Accessed July 1, 2025.« Back to 2019 International Congress
MDS Abstracts - https://www.mdsabstracts.org/abstract/normative-curves-for-selected-unified-huntington-disease-rating-scale-measures/